Our Clinical Trials
At Valley Eye Physicians & Surgeons, our team participates in national clinical research to help advance the understanding and treatment of complex eye conditions. By joining these studies, eligible patients may gain access to new therapies while contributing to the development of future treatments that help improve vision and quality of life.
If you’re interested in participating in one of our clinical trials, please speak with our team to determine your eligibility.
Active Clinical Trials
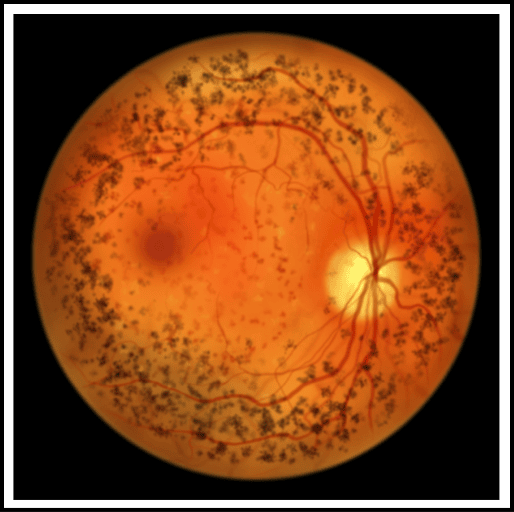
Protocol AF — Diabetic Retinopathy (DR)
Phase III study sponsored by the NEI’s DRCR Network and Kowa Research Institute
PROMINENT-Eye Ancillary Study: Diabetic Retinopathy Outcomes in a Randomized Trial of Pemafibrate versus Placebo
This study examines whether pemafibrate can help improve diabetic retinopathy, a condition caused by damage to the retinal blood vessels from high blood sugar. Participants must already be enrolled in the main PROMINENT study, which is investigating pemafibrate’s role in reducing heart and vascular complications.
Main Criteria:
- Participation in the main PROMINENT study
- Diagnosed diabetic retinopathy in at least one eye
Closed Clinical Trials
Protocol AC — Diabetic Macular Edema (DME)
Phase III study sponsored by the National Eye Institute’s DRCR Network
Aflibercept versus Bevacizumab with Deferred Aflibercept for DME
This study compares the effectiveness of Eylea (Aflibercept) and Avastin (Bevacizumab) in treating swelling in the center of the retina, known as diabetic macular edema. The goal is to determine whether starting treatment with Eylea provides better outcomes than beginning with Avastin and switching to Eylea only if needed.
Main Criteria:
- Vision of 20/50 or worse
- Diabetic macular edema meeting specific criteria in at least one eye
- No treatment for DME within 4 months prior to starting the study
- No recent or anticipated retinal laser therapy within 6 months
- Not currently on dialysis or with a history of kidney transplant
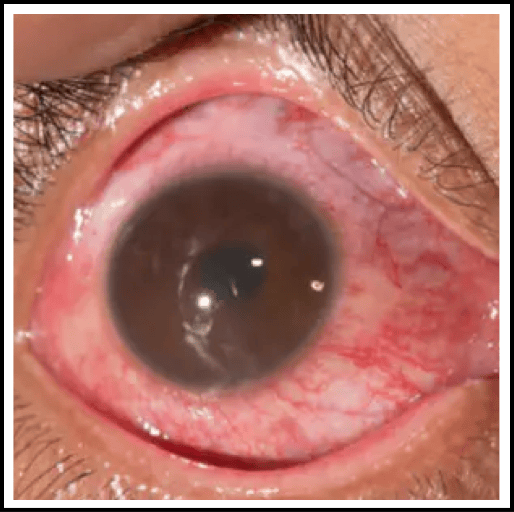
Anterior Uveitis Study
Phase III study sponsored by Aldeyra Therapeutics, Inc.
A Phase 3 Randomized, Double-Masked, Vehicle-Controlled Trial of ADX-102 Ophthalmic Solution
This study evaluates the safety and efficacy of ADX-102 ophthalmic solution in patients with non-infectious anterior uveitis. Up to 100 patients will be enrolled and randomly assigned to receive either ADX-102 or a placebo treatment over 4 weeks. The primary goal is to measure the reduction of inflammation in the front of the eye.
Main Criteria:
- 18 years of age or older
- Subjects with acute non-infectious anterior uveitis with onset of symptoms within the previous 2 weeks.
- Best corrected visual acuity (BCVA) better than or equal to 35 letters in the study eye and 65 letters in the non-study eye using ETDRS testing.
- NO severe/serious ocular pathology in the study eye(s)
- NO active intermediate or posterior uveitis in the study eye(s).
NO previous anterior uveitis episode in the study eye, less than or equal to 4 weeks prior to screening
Protocol AB — Vitreous Hemorrhage from Proliferative Diabetic Retinopathy (PDR)
Phase III study sponsored by the NEI’s DRCR Network
Intravitreous Anti-VEGF vs. Prompt Vitrectomy for Vitreous Hemorrhage from PDR
The objectives of this study are to 1) evaluate and compare visual acuity outcomes over 2 years of a prompt vitrectomy + panretinal photocoagulation (PRP) regimen and an intravitreous aflibercept regimen in eyes with VH from PDR for which intervention is deemed necessary, and 2) characterize the follow-up course for the 2 treatment regimens, including but not limited to post-operative complications for the vitrectomy group, and number of injections needed and percent requiring vitrectomy in the intravitreous aflibercept group.
Major Criteria:
- Immediate vitrectomy not required (investigator and participant are willing to wait at least 4 months)
- Visual acuity letter score less than or equal to 78 (approximate Snellen equivalent 20/32)
- No systemic anti-vascular endothelial growth factor or pro-vascular endothelial growth factor treatment within 4 months prior to randomization
- No retinal detachment
- No diabetic macular edema
- No history of vitrectomy
Protocol W (Phase III study sponsored by NEI’s DRCR Network) on Diabetic Retinopathy (DR)
Intravitreous Anti-VEGF Treatment for Prevention of Vision-Threatening Diabetic Retinopathy in Eyes at High Risk
This randomized study tests whether intravitreous anti-VEGF aflibercept (Eylea) given to diabetic patients before progression to retinal neovascularization or proliferative diabetic retinopathy (PDR) decreases the risk of DR worsening. Subjects will be randomized to take either aflibercept (Eylea) or standard of care (sham injections). If this study demonstrates that intravitreous aflibercept treatment is effective and safe for reducing the incidence of PDR or center-involved DME (CI-DME) in eyes at high risk for these complications, it will provide a new strategy to prevent vision-threatening complications of diabetes for patients. Applying intravitreous aflibercept earlier in the course of disease (i.e., at baseline severe non-proliferative diabetic retinopathy) could reduce future treatment burden while potentially yielding similar or better long-term visual outcomes if PDR and DME are prevented.
Major Criteria:
- Severe NPDR (ETDRS level 53) according to investigator 4-2-1 rule
- Severe hemorrhages in at least 4 quadrants, or
- Definite venous beading in at least 2 quadrants, or
- Moderate IRMA in at least 1 quadrant
- Vision 20/25 or better
- No center-involved DME on OCT
- No history of DME/DR treatment in the prior 12 months and <4 prior injections at any time
- No prior PRP
Completed Clinical Trials
PEACHTREE protocol CLS1001-301 (Phase III study sponsored by Clearside Biomedical)
Non-infectious uveitis trial
This randomized study evaluates the safety and efficacy of suprachoroidally administered triamcinolone acetonide, CLS-TA, in subjects with macular edema associated with non-infectious uveitis. The primary outcome measure is the improvement of visual acuity in the study eye.
CEDAR protocol 150998-005 (Phase III study sponsored by Allergan) on wet AMD
Safety and Efficacy of Abicipar Pegol (AGN-150998) in Patients with Neovascular Age-related Macular Degeneration
This randomized study tests the investigational product, abicipar, administered at baseline (day 1), weeks 4 and 8, followed by doses at 8-week or 12-week intervals through week 96, compared with 0.5 mg ranibizumab (Lucentis) administered every 4 weeks through week 100. The primary outcome measure is the improvement of visual acuity in the study eye. This investigational medication may reduce the frequency of intravitreous injections compared to monthly injections of ranibizumab.
ORBIT protocol TG-MV-018 (Phase IV study sponsored by ThromboGenics, Inc.) on VMA
Ocriplasmin Research to Better Inform Treatment (ORBIT)
This is an observational study to record clinical outcomes and safety in patients receiving JETREA® for the treatment of symptomatic vitreomacular adhesion (VMA) as per standard of care in US retina clinics.
SARIL-NIU-SATURN protocol ACT13480 (Phase III study sponsored by Sanofi) on Posterior Uveitis
A randomized, double-masked, and placebo-controlled study to evaluate the efficacy and safety of sarilumab administered subcutaneously every 2 weeks in patients with Non-infectious, Intermediate, Posterior, or Pan-Uveitis (NIU) Sarilumab (SAR153191)
This is a randomized study where the primary objective is to evaluate the efficacy of 200 mg sarilumab q2w at W16 in patients with NIU. The goal of NIU treatment is to control inflammation, prevent visual loss, and minimize progression.
NS2-02 (Phase II study sponsor Aldeyra Therapeutics, Inc.) on Anterior Uveitis
A Phase 2 Randomized, Investigator-Masked, Comparator-controlled Trial to Evaluate the Safety and Efficacy of NS2 Eye Drops in Patients with Anterior Uveitis
The investigational medication NS2, a small-molecule aldehyde trap, may reduce inflammation by lowering aldehyde levels. Free aldehydes are thought to be associated with inflammatory diseases, including uveitis. NS2 is a non-steroidal that does not carry the same risks of prolonged corticosteroid use. The purpose of this study is to determine whether NS2 is safe and effective in the treatment of uveitis compared to the FDA-approved steroid eye drop, Pred Forte®.
Learn More About Clinical Trials
Participation in clinical research is voluntary and closely monitored. Each study follows strict safety standards established by the National Eye Institute (NEI) and the American Academy of Ophthalmology.
For more information about eligibility or active enrollment, please contact our research team.
Peer-Reviewed Publications
- Velez G, Weingarden AR, Lei H, Kazlauskas A, Gao G. SU9518 inhibits proliferative vitreoretinopathy in fibroblast and genetically modified Müller cell- induced rabbit models. Invest Ophthalmol Vis Sci. 2013 Feb 19;54(2):1392-7. doi10.1167/iovs.12-10320 PMID: 23341018
- Velez G, Weingarden AR, Tucker BA, Lei H, Kazlauskas A, Young MJ. Retinal pigment epithelium and Muller progenitor cell interaction increase Müller progenitor cell expression of PDGFR and ability to induce proliferative vitreoretinopathy in a rabbit model. Stem Cells Int 2012;2012:106486. Epub 2012 Aug 23. PMID: 22966235
- Lei H, Rheaume MA, Velez G, Mukai S, Kazlauskas A. Expression of PDGFR is a determinant of the PVR potential of ARPE19 cells. Invest Ophthalmol Vis Sci August 2011;52: 5016-5021. PMID: 21642621
- Lei H, Velez G, Cui J, Matsubara J, Kazlauskas A. Pathologic signaling of PDGFR alpha involves chronic activation of Akt and suppression of p53. Mol Cell Biol 2011 May; 31(9):1788-99. Epub 2011 Feb28. PMID: 21357737
- Lei H, Velez G, Cui J, Samad A, Maberley D, Matsubara J, Kazlauskas A. N-Acetylcysteine suppresses retinal detachment in an experimental model of PVR. Am J Pathol 2010 Jul:177(1):132-40. Epub 2010 May 20. PMID: 20489144
- Lei H, Velez G, Hovland P, Hirose T, Gilbertson D, Kazlauskas A. Growth factors outside of the PDGF family drive experimental PVR. Invest Ophthalmol Vis Sci 2009 July; 50(7):3394-403. PMID: 19324843
- Lei H, Velez G, Hovland P, Hirose T, Kazlauskas A. Plasmin is the major protease responsible for processing PDGF-C in the vitreous of patients with proliferative vitreoretinopathy. Invest Ophthalmol Vis Sci 2008 Jan; 49(1): 42-8. PMID: 18172073
- Lei H, Hovland P, Velez G, Haran A, Gilbertson D, Hirose T, Kazlauskas A. A potential role for PDGF-C in experimental and clinical proliferative vitreoretinopathy. Invest Ophthalmol Vis Sci 2007 May; 48(5):2335-42. PMID: 17460299
- Buggage RR, Levy-Clarke G, Sen HN, Ursea R, Srivastava SK, Suhler EB, Altemare C, Velez G, Ragheb J, Chan CC, Nussenblatt RB, Bamji AT, Sran P, Waldmann T, Thompson DJ. A double-masked, randomized study to investigate the safety and efficacy of daclizumab to treat the ocular complications related to Behcet’s disease. Ocul Immunol Inflamm. 2007 Mar-Apr;15(2):63-70. PMID: 17558830
- Velez G, Kansupada K, Csaky K, Reed GF, Whitcup SM, Nussenblatt RB. Serous Retinal Detachment in Posterior Uveitis. Ann Ophthalmol 2004; 36(2):103-110.
- Nussenblatt RB, Thompson DJ, Li Z, Chan CC, Peterson JS, Robinson RR, Shames RS, Nagarajan S, Tang MT, Mailman M, Velez G, Roy C, Levy-Clarke GA, Suhler EB, Djalilian A, Sen HN, Al-Khatib S, Ursea R, Srivastava S, Bamji A, Mellow S, Sran P, Waldmann TA, Buggage RR. Humanized anti-interleukin-2 (IL-2) receptor alpha therapy: long-term results in uveitis patients and preliminary safety and activity data for establishing parameters for subcutaneous administration. J Autoimmun. 2003 Nov; 21(3):283-93. PMID: 14599854
- Velez G, Weiter JJ. Cataract extraction and age-related macular degeneration: associations, diagnosis and management. Seminars in Ophthalmology 2002 Sep-Dec; 17(3-4):187-195. PMID: 12759849
- Velez G, Boldt HC, Whitcup SM, Nussenblatt RB, Robinson MR. Local methotrexate and dexamethasone phosphate for recurrent primary intraocular lymphoma. Ophthal Surg and Lasers 2002; 33:329-333. PMID: 12134997
- Velez G, Chan CC, Csaky KG. Fluorescein angiographic findings in primary intraocular lymphoma. Retina 2002; 22:37-43. PMID: 11884876
- Velez G, Yuan P, Sung C, Tansey G, Reed GF, Chan CC, Nussenblatt RB, Robinson MR. Pharmacokinetics and toxicity of intravitreal chemotherapy for primary intraocular lymphoma. Arch Ophthalmol 2001; 119:1518-1524. PMID: 11594954
- Velez G, Buggage RR. Interleukin-10 and intraocular-central nervous system lymphoma. Ophthalmology 2001; 108(3):427-428. PMID: 11237884
- Velez G, Roy CE, Whitcup SM, Chan CC, Robinson MR. High dose intravitreal ganciclovir and foscarnet for cytomegalovirus retinitis. Am J Ophthalmol 2001; 131(3):396-397. PMID: 11239885
- Velez G, de Smet MD, Whitcup SM, Robinson MR, Nussenblatt RB, Chan CC. Iris involvement in primary intraocular lymphoma: report of two cases and review of the literature. Surv Ophthalmol 2000; 44:518-526. PMID: 10906383
- Velez G, Whitcup SM. New developments in sustained release drug delivery for the treatment of intraocular disease. Br J Ophthalmol 1999; 83:1225-1229. PMID: 10535845
- Buggage RR, Velez G, Myers-Powell B, Shen D, Whitcup SM, Chan CC. Primary intraocular lymphoma with a low interleukin 10 to interleukin 6 ratio and heterogeneous IgH gene rearrangement. Arch Ophthalmol 1999; 117:1239-42. PMID: 10496399
Books and Chapters
- Velez G, Whitcup SM. Ophthalmologic Manifestations. in Vasculitis. Ed Gene V. Ball, S. Louis Bridges (Oxford University Press Inc, New York, NY; 2001), p. 80-101.
Presentations and Abstracts
- Velez G. A study of geographic atrophy as the footprint of choroidal neovascularization in age-related macular degeneration. 2017. Association for Research in Vision and Ophthalmology abstract 2331
- Velez G. De novo and recurrent polypoidal lesions are amenable to anti-VEGF therapy. 2016. Association for Research in Vision and Ophthalmology abstract 4964
- Velez G. Chlorhexidine gluconate antisepsis for intravitreal injection. 2015. Association for Research in Vision and Ophthalmology abstract 4179
- Velez G. Intravitreal triamcinolone acetonide with anti-VEGF therapy for the management of polypoidal choroidal vasculopathy. 2014. Association for Research in Vision and Ophthalmology abstract 3905
- Velez G. Ranibizumab for the treatment of diabetic macular edema in patients treated with Bevacizumab. 2013. Association for research in Vision and Ophthalmology abstract 2395
- Velez G, Roy A. Surface substrates affect the behavior and survival of Müller glia derived stem cells. 2012. Association for Research in Vision and Ophthalmology abstract 5917
- Velez G, Lei H, Kazlauskas A, Kaushal S, Young M. SU9518 inhibits proliferative vitreoretinopathy (PVR) in a Muller cell rabbit model of PVR. 2011. Association for Research in Vision and Ophthalmology abstract 2065.
- Lei H, Rheaume MA, Velez G, Mukai S, Kazlauskas A. Expression of PDGFRa in retinal pigment epithelial cells promotes experimental proliferative vitreoretinopathy. 2011. Association for Research in Vision and Ophthalmology abstract 6590.
- Velez G, Kazlauskas A, Young M. SU 9518 inhibits proliferative vitreoretinopathy (PVR) in a rabbit model. 2010. Association for Research in Vision and Ophthalmology abstract 6086.
- McRee SK, Tucker B, Bradbury MJ, Young M, Velez G. Indocyanine green (ICG) induces increased Muller cell expression of adhesion molecules and membrane metalloproteinases in vitro. 2010. Association for Research in Vision and Ophthalmology abstract 5762.
- Lei H., Velez G, Kazlauskas A. Indirect activation of PDGFR alpha engages a PI3K/AKT/p53 dependent pathway that promotes cellular responses intrinsic to PVR. 2010. Association for Research in Vision and Ophthalmology abstract 6084.
- Velez G, Tucker BA, Weingarden AR, Young, MJ. Mueller cell de-differentiation leads to increased expression of platelet-derived growth factor receptor and retinal pigment epithelium cell markers. 2009. Association for Research in Vision and Ophthalmology abstract 1295
- Bradbury MJ, Velez G. Combined intravitreal bevacizumab and photodynamic therapy for the treatment of very resistant choroidal neovascularization in age-related macular degeneration. 2009. Association for Research in Vision and Ophthalmology abstract 1915
- Weingarden AR, Tucker BA, Lei H, Young MJ, Velez G. Upregulation of platelet-derived growth factor receptor alpha in RPE and Muller cell co-cultures. 2009. Association for Research in Vision and Ophthalmology abstract 2707
- Kazlauskas A, Lei H, Velez G. Growth factors outside of the PDGF family drive experimental PVR. 2009. Association for Research in Vision and Ophthalmology abstract 6227
- Lei H, Velez G, Kazlauskas A. The role of growth factors outside of the PDGF family in experimental proliferative vitreoretinopathy. 2008. Association for Research in Vision and Ophthalmology abstract 4896
- Velez G, Bradbury MJ. Combined intravitreal bevacizumab (Avastin) and triamcinolone acetonide (Kenalog) for the treatment of choroidal neovascularization (CNV) associated with age-related macular degeneration (ARMD). 2008. Association for Research in Vision and Ophthalmology abstract 558
- Velez G, Lei H, Kazlauskas A. Comparison of fibroblasts and RPE cells for their ability to induce PVR in rabbits. 2007. Association for Research in Vision and Ophthalmology abstract 5778
- Bradbury M, Velez G. Intravitreal Avastin vs. laser photocoagulation for the treatment of peripapillary choroidal neovascularization. 2007. Association for Research in Vision and Ophthalmology abstract 1460
- Velez G, Lei H, Kazlauskas A. Intravitreal STI571 inhibition of proliferative vitreoretinopathy in an experimental rabbit model is dose dependent. 2006. Association for Research in Vision and Ophthalmology abstract 4685
- Lei H, Hovland P, Velez G, Eriksson U, Gilbertson DG, Hirose T, Kazlauskas A. A potential role for PDGF-CC in proliferative vitreoretinopathy. 2006. Association for Research in Vision and Ophthalmology Abstract 3828
- Velez G, Kazlauskas A. Intravitreal STI571 inhibits development of proliferative vitreoretinopathy in an experimental rabbit model. 2005. Association for Research in Vision and Ophthalmology abstract 5519
- Velez G, Weiter JJ. Assessment and comparison of risk factors in subretinal hemorrhages associated with choroidal neovascularization and suprachoroidal hemorrhages. 2003. Association for Research in Vision and Ophthalmology abstract 4921
- Velez G, Silk W, Csaky KG. Correlation of in-vivo fluorescein angiography findings with in-vitro fluorescein uptake by monocytes. 2002. Association for Research in Vision and Ophthalmology abstract 516
- Velez G, Baudo TA, Luu RP, Murphy RP, Glaser BM. High Speed ICG Angiographic characterization of feeder vessels to choroidal neovascularization associated with ocular inflammatory disease. Invest Ophthalmol Vis Science 2001; 42(4): S230. Abstract 1240
- Glaser BM, Baudo TA, Velez G, Luu JK, Murphy RP. Feeder vessel treatment for age-related macular degeneration with classic choroidal neovascularization (CNV). Invest Ophthalmol Vis Science 2001; 42(4): S231. Abstract 1243
- Luu JK, Baudo TA, Velez G, Murphy RP, Glaser BM. Internal tamponade without tPA for the treatment of submacular hemorrhage in age-related macular degeneration. Invest Ophthalmol Vis Science 2001; 42(4): S234. Abstract 1264
- Baudo TA, Velez G, Luu JK, Murphy RP, Glaser BM. Incidence of predominantly classic choroidal neovascular membranes (CNV) in exudative age related macular degeneration (AMD): percentage of new patients eligible for photodynamic therapy with Vereteporfin. Invest Ophthalmol Vis Science 2001; 42(4): S438. Abstract 2360
- Velez G, Kansupada K, Csaky KG, Whitcup SM, Nussenblatt RB. Serous retinal detachment in patients with uveitis. AAO, 2000
- Velez G, King BA, Yuan P, Whitcup SM, Robinson MR. Thalidomide analogue (EM138) sustained- release implants to treat choroidal neovascularization associated with macular degeneration. Invest Ophthalmol Vis Science 2000;41(4): S180.
- Buggage RR, Velez G, Chan CC, Smith JA. New keratoconjunctival findings in HTLV-1 infection. American Uveitis Society Meeting, AAO, 1999.
- Velez G, Robinson MR, Durbin T, et al. Thalidomide sustained release devices for choroidal neovascularization: an in-vitro analysis. Invest Ophthalmol Vis Science 1999; 40(4): S448
- Brusie SR, Velez G, Wagner DG, Cupples HP. Epidemiology and long-term outcomes of open-globe missile injuries among children. Invest Ophthalmol Vis Science 1999; 40(4): S168
- Velez G, Wagner DG. The role of scleral buckle in the management of open globe injuries. Invest Ophthalmol Vis Science 1998; 39(4): Abstract 4648
- Bonsall D, Territo J, Velez G, Chrousos G. Vertical Strabismus after cataract surgery. AAPOS, 1998
- Velez G, Wagner DG. Epidemiology of open globe injuries and the elderly. Invest Ophthalmol Vis Science 1997;38(4): S282
Putting Your Eye Health First
We provide comprehensive eye care with a focus on medical and surgical treatments for complex eye conditions. Our experienced team combines experience with compassionate care to deliver personalized treatment tailored to each patient’s needs.
In addition to routine eye care, we have the resources to offer a high standard of innovative treatments, including cataract management, glaucoma care, and specialized retina and uveitis services. Using modern technology and a patient-focused approach, we strive to provide effective care and positive results.
Our mission is to provide our community with exceptional, personalized eye care. Your eye health is our priority—schedule your appointment with us today.
Visit Our Locations in Massachusetts
Ayer Location
We are on Groton Road, and parking is available outside the clinic. We look forward to welcoming you!
- 978-772-4000
- 979-772-3066
- 190 Groton Rd., Suite 240
- Ayer, MA 01432
Our Hours
Marlboro Location
You can find us across from the Marlboro Animal Hospital. Contact us to schedule your appointment today!
- 508-496-9000
- 508-486-9001
- 420 Lakeside Ave., Suite 103
- Marlboro, MA 01752
Our Hours
Worcester Location
We are on Eastern Avenue, and parking is available around the clinic. Our team is looking forward to seeing you!
- 508-552-0066
- 508-753-1677
- 2 Eastern Ave
- Worcester, MA 01613
Our Hours